 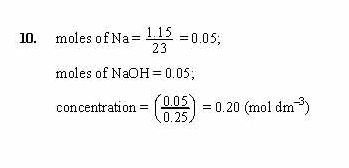
Thus, its molar mass involves: MNa 22.989 g/mol. This is a long problem, so I certainly would check the arithmetic. The molecular molar mass consists of the atomic masses of each atom. The balanced chemical equation for the reaction between the sodium metal and the water is as follows- Na (s) + HO (l) NaOH (aq) + ½. #"Volume of NaOH titrant required"#=#(2xx1.614xx10^-3*mol)/(7.158xx10^-2*mol*L^-1)xx1000*mL*L^-1=?*mL#.Īnd thus, we require under #50*mL# of titrant, and this is a good volume for a titration. Number of moles of sodium hydroxide present mass of sodium hydroxide/molar mass of sodium hydroxide 10.40 g/(40 g/mol) 0.26 mol. Match the words in the left column to the. Complete the following sentences regarding the concentration of the prepared solution. #"Moles of oxalic acid"#=#(0.1453*g)/(90.03*g*mol^-1)=1.614xx10^-3*mol#. Transcribed image text: You need to prepare a 2.40 Msolution of sodium hydroxide (molar mass of sodium hydroxide 40.00 g/mol), but you only have a 10 mL graduated cylinder and a 25 mL beaker. #HO_2C-CO_2H*(OH_2)_2#, so if your results are off, question the quality of your oxalic acid, and you can assess this easily by melting point).Īgain, we need a stoichiometric equation: #HO_2C-CO_2H#, with a molecular mass of #90.03*g*mol^-1# (the dihydrate is much common, i.e. So we have #NaOH#, and we use to titrate a mass of oxalic acid. Given the stoichiometry, this molar quantity was contained in the volume of titrant, i.e. #C_6H_4(CO_2^(-)K^(+))(CO_2H) + NaOH rarr C_6H_4(CO_2^(-)K^(+))(CO_2^(-)Na^(+))+H_2O#Īnd this is a convienient 1:1 stoichiometry, so moles of phthalate is equivalent to moles of base:Īnd (ii) equivalent quantities of #"KHP"#, 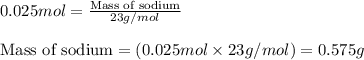
First we need to calculate the molar mass (molecular weight) of H2O2 (hydrogen peroxide) so we know how. We need (i) a stoichiometrically balanced equation for our primary standard: 17.0 grams are given in the text of the problem.
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
